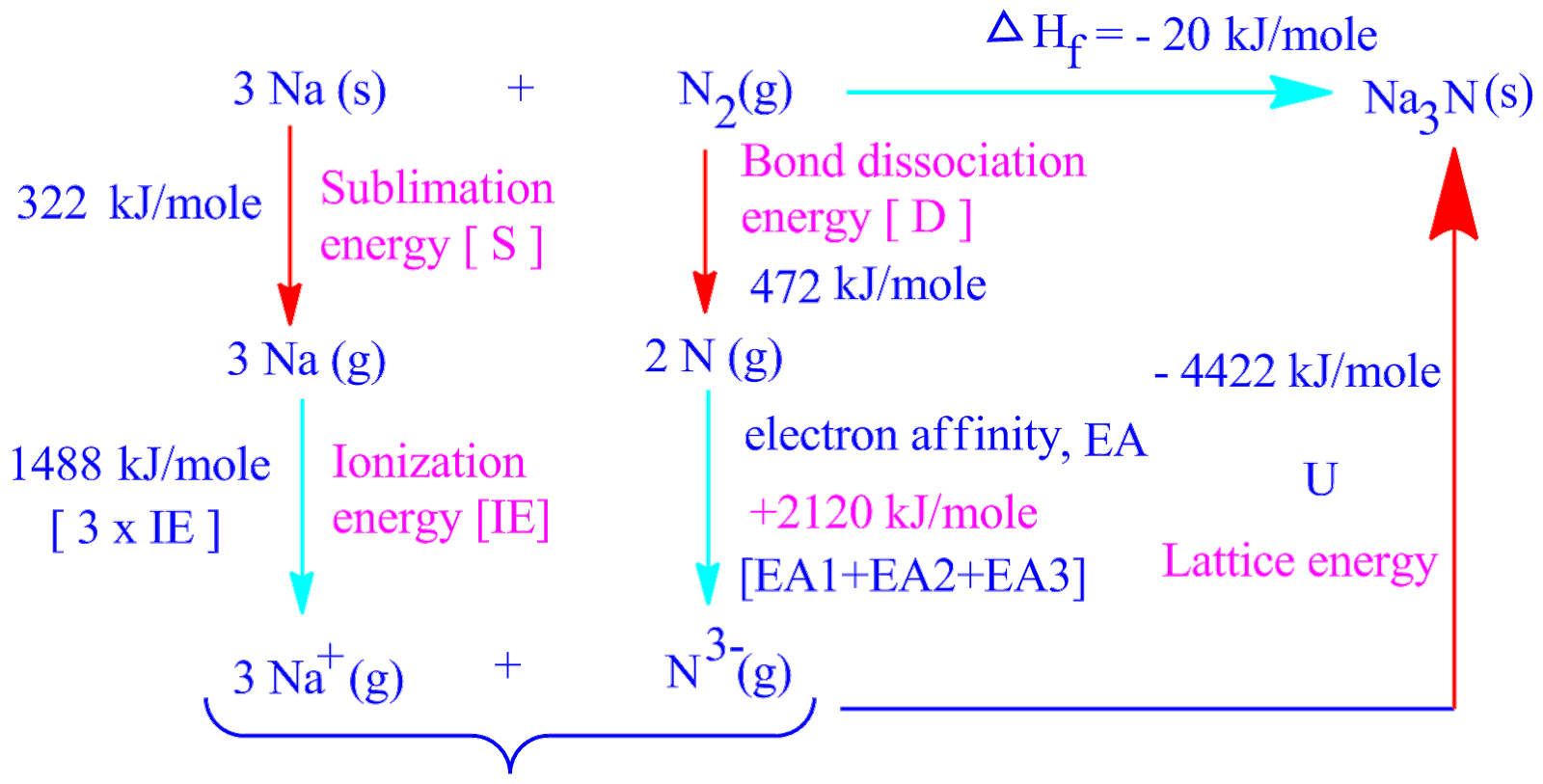
The formation of sodium chloride occurs when sodium ions interact with chloride ions. The ionisation potential (+IP) is the amount of energy required.Ĭhlorine dissociation: The energy required to dissociate a chlorine molecule into two chlorine atoms is the dissociation energy (+D/2).Ĭhloride ion formation: To produce chloride ion, the Cl atom takes an electron. The ionisation of sodium occurs when an electron is removed from the sodium atom, resulting in the formation of a sodium ion. The sublimation energy (+S) is necessary for this to happen. Sublimation of sodium: Sublimation is the process by which solid sodium becomes gaseous sodium. Method of indirection: Several processes are involved in the production of sodium chloride in this method: There are two ways to make sodium chloride:ĭirect Method: Sodium chloride is formed when metallic sodium combines with chlorine gas. It should be emphasised that this law also applies to cyclic processes.Ĭalculation of Lattice Energy of Nacl Using Born Haber Equation Then +Q = q 1 + q 2 + q 3 according to Hess' law. 
For example, the heat of reaction (H) = +Q for a chemical reaction given byĪlternatively, if the reaction occurs in multiple steps: According to Hess' rules, whether a chemical reaction occurs in one or more steps, the overall heat of the reaction remains constant. Hess' law of constant heat of summation underpins the Born-Haber cycle. Calculation of Lattice Energy Using Born Haber Cycle As a result, the ionic solid's lattice energy increases as the size of the constituent ions decreases. 
The ionic lattice of smaller atoms has fewer interatomic distances and stronger binding forces. The weaker the electrostatic forces binding ions together in a lattice, the lower the lattice energy. The distance between the ions is inversely related to the lattice energy of an ionic molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
